Metal minerals scarcity:
A call for managed austerity and the elements of hope
Dr. A.M.
Diederen, MSc.
TNO
Defence, Security and Safety
andre.diederen@tno.nl
March
10, 2009
[From theoildrum.com, http://europe.theoildrum.com/node/5239#more]
Abstract
If we keep following the ruling
paradigm of sustained global economic growth, we will soon run out of cheap and
plentiful metal minerals of most types. Their extraction rates will no longer
follow demand. The looming metal minerals crisis is being caused primarily by
the unfolding energy crisis. Conventional mitigation strategies including
recycling and substitution are necessary but insufficient without a different
way of managing our world’s resources. The stakes are too high to gamble on
timely and adequate future technological breakthroughs to solve our problems.
The precautionary principle urges us to take immediate action to prevent or at
least postpone future shortages. As soon as possible we should impose a co-ordinated
policy of managed austerity, not only to address metal minerals shortages but
other interrelated resource constraints (energy, water, food) as well. The
framework of managed austerity enables a transition towards application
(wherever possible) of the ‘elements of hope’: the most abundant metal (and
non-metal) elements. In this way we can save the many critical metal elements
for essential applications where complete substitution with the elements of
hope is not viable. We call for a transition from growth in tangible
possessions and instant, short-lived luxuries towards growth in consciousness,
meaning and sense of purpose, connection with nature and reality and good
stewardship for the sake of next generations.
Introducing metal minerals scarcity
and managed austerity
Undoubtedly, the global economic
growth of the last century, fuelled by and accompanied by exponential growth in
population and consumption of resources like fossil fuels, water, food and
metal minerals, is unsustainable. Now that we are nearing the second decade of
the 21st century, we are beginning to notice the consequences of
supply gaps of various resources. This paper focuses on the issue of metal
minerals scarcity within the constellation of interconnected problems of scarcity
of water and food, pollution and climate change and most notably scarcity of
energy. In case of unlimited energy supply, metal minerals extraction would
only be limited by the total amount of mineral resources. However, due to the
scarcity of energy, the extraction rates of most types of metal minerals will
cease to follow demand. Probably the only acceptable long-term solution to
avoid a global systemic collapse of industrial society, caused by these
resource constraints, is a path towards managed austerity. Managed austerity
will have to be a combination of changes in technology and changes in both
individual and collective human behaviour. Managed austerity could prevent
non-desirable ‘solutions’ by doing much too little much too late (also known as
‘business as usual’) which could ultimately result in large scale conflicts,
global chaos and mass starvation of the world’s population.
Energy scarcity
Humanity has depleted a significant
part of its inheritance of highly concentrated energy resources in the form of
fossil fuels. Although huge quantities of these resources remain untapped, the
worldwide extraction rate (production flow) has reached a plateau and will soon
begin to decline [1,2,3,4,5,6]. The result is an ever
widening supply gap because sustained global economic growth requires sustained
growth in available energy. Figure 1 gives the general depletion picture for
oil and gas [1] in giga barrels of oil equivalent (Gboe) and the left part of
the bell-shaped curve strongly resembles a logistic curve. The initial stage of
growth is approximately exponential, growth slows as
saturation begins (‘the low-hanging fruit has been picked’) and at maturity
growth stops and a maximum is reached. The maximum production rate is referred
to as the ‘peak’ and is not a sharp deflection point in the curve but rather a
plateau region.
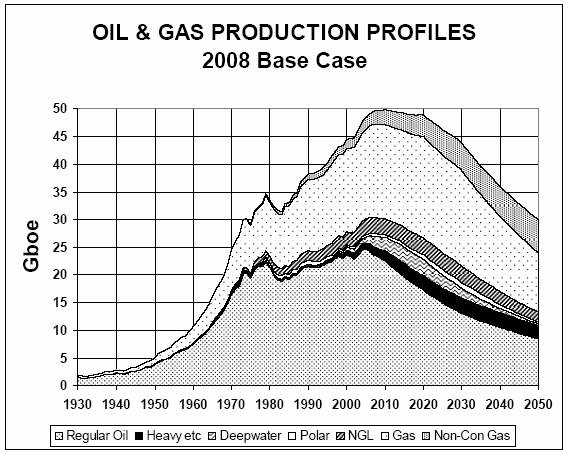
Figure 1:
Depletion curve for oil and gas [1]
It is important to realise that the
peak date in the depletion graph (figure 1) is not the same as the half date
because production can continue for a long period after the peak. The actual
depletion curve will presumably be asymmetric, having a peak date before the
half date. Although the exact peak date for oil and gas is being contested
(ranging from 2005 to somewhere during the next few decades), experts and
authorities seem to converge on a peak date within the next few years. Oil and
gas are currently the world’s most important energy sources. Transportation for
instance is currently almost entirely dependent on oil. Coal will not be able
to fill the energy gap after the peak in oil and gas. According to [7] coal may
peak around 2025. Again, this does not imply exhaustion of coal reserves, it is
quite possible that more coal will be left for extraction after the peak date
than has been extracted in total in the years before. The crucial point is that
a maximum production rate will be reached after which supply can
no longer follow demand. It is estimated that oil, gas and coal combined
will reach their ‘peak all fossil fuels’ close to 2020 [8]. All other energy
resources combined (nuclear, hydro, wind, solar, biofuels, tidal, geothermal
and so on) cannot fill the supply gap in time [9,10,11,12].
Timely and massive utilisation of these other energy resources is limited by
various constraints like lack of concentration, intermittency, issues related
to conversion and storage and last but not least the required massive input of fossil
fuels and metal minerals. Therefore we will probably be confronted with a peak
in global energy production within the next 10 to 15 years, despite progress in
technology.
Metal minerals scarcity
The depletion graphs of most metal
minerals will resemble the curve for oil and gas (figure 1). Figure 2 gives an
example for zirconium mineral concentrates [13].
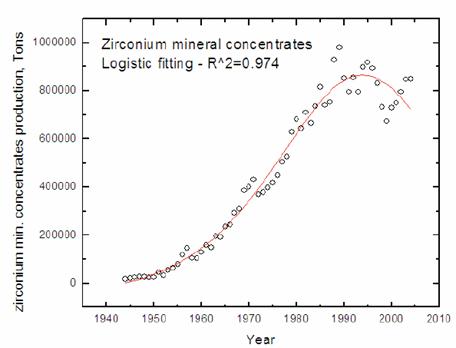
Figure 2:
Depletion curve for zirconium mineral concentrates [13]
Many warnings in the past of
impending metal minerals shortages have been proven wrong because of the
availability of cheap and abundant fossil fuels. Every time the ratio of
reserves to production of a certain metal mineral became uncomfortably small,
the reserves of that mineral were being revised upwards because it became
economically feasible to extract metals from the so-called reserve base or
resource base. Reserves are defined as those ores that can be economically extracted
at the time of determination and the term reserves need not signify that
extraction facilities are in place and operative. The decades-old paradigm
which states that reserves will be revised upwards (to include lower ore
grades) as soon as supply gaps are looming, is no longer valid without cheap
and abundant energy. Mining and extraction (concentration) consume huge amounts
of energy. The energy required for extraction grows exponentially with lower
ore grades. This is illustrated in figure 3 for iron ore and aluminium ore
[14]. The highest ore grades have already been depleted or are already being
mined. Because of energy constraints, the largest parts of mineral deposits are
out of reach for economically viable exploitation, see figure 4 [15].
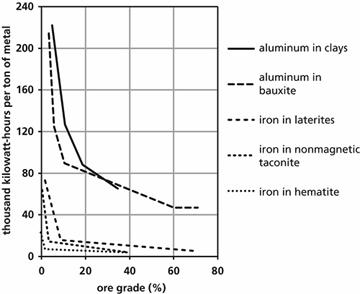
Figure 3:
Relation between required energy for extraction and ore grade [14]
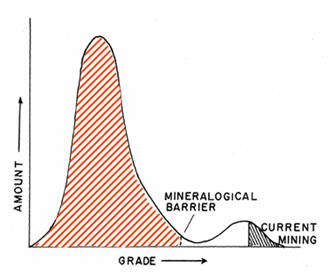
Figure 4: Mineralogical barrier for
most elements [15]
Below the so-called mineralogical
barrier (the red shaded area in figure 4), one would
essentially have to pull the rock chemically apart to extract all individual
elements. This is of course prohibitively energy intensive. For this reason it
is very doubtful that meaningful parts of the reserve base or resource base of
many metal minerals will ever be upgraded to reserves [16]. It is even
questionable whether all currently stated reserves are fully exploitable given
the ever growing constraints with regard to energy required [13].
The trend of geologically and
physically based minerals scarcity will be further enhanced by other factors.
Global (‘average’) shortages will most likely be preceded by spot shortages
because of geopolitics and export restrictions, as many important metal
minerals are concentrated in just a few countries, often outside the western
industrialized world (e.g. China).
Extraction rates and reserves of
metal minerals
Known data of extraction and
consumption rates of metal minerals and their reserves indicate that the
so-called ‘peak production’ for most metal elements will lie in the near
future. The data from table 1 and figures 5 through 9 support this statement.
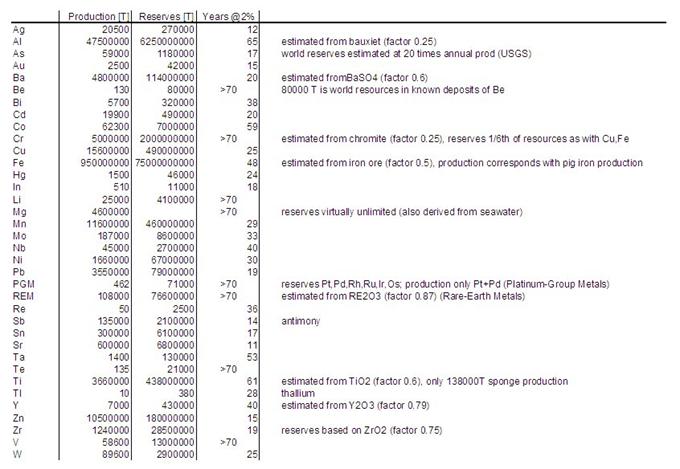
Table 1 represents an overview
presented by the US Geological Survey [17] of global annual primary production
and global reserves of a large number of metal minerals. Their production goes
into various products and compounds, part of them being steels, alloys and
metal products. The remaining ‘lifetimes’ are calculated based on a modest
consumption growth of 2% per year. The elements predicted to have a ‘lifetime’
of less than 50 years are summarized in figure 5. Of course, these minerals are
not completely depleted in this period, but their peak production lies well
before the estimated moment. Compare the result for zirconium with figure 2:
the remaining ‘lifetime’ of zirconium is 19 years and the peak date is already
behind us (1994). Although exact data fail, the elements strontium through
niobium (of figure 5) will soon reach their peak production or have already
passed their maximum extraction rates.
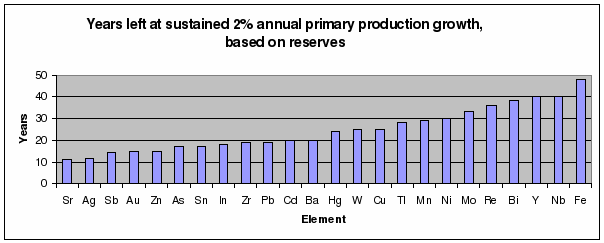
Figure 5: Years left of reserves at
a sustained annual global primary
production growth of 2% (based on table 1)
Figure 6 through 9 depict in more
detail global annual production rates and the known reserves. The annual primary
production of iron dwarfs all other metal elements combined. Despite its huge
reserves, iron will last less than 3 generations (less than 50 years) as far as
cheap and abundant primary production is concerned, due to the enormous scale
of its annual global consumption. The only viable long-term alternative to iron
and in fact all metals at this scale of consumption would be magnesium.
Magnesium reserves are virtually unlimited because of its abundance and
associated accessibility in seawater [20].
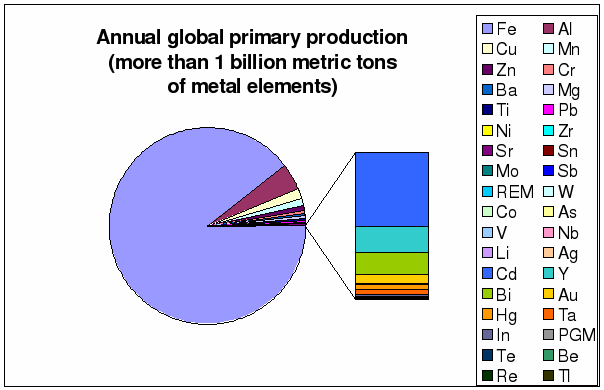
Figure 6: Distribution of annual
global primary production (based on table 1)
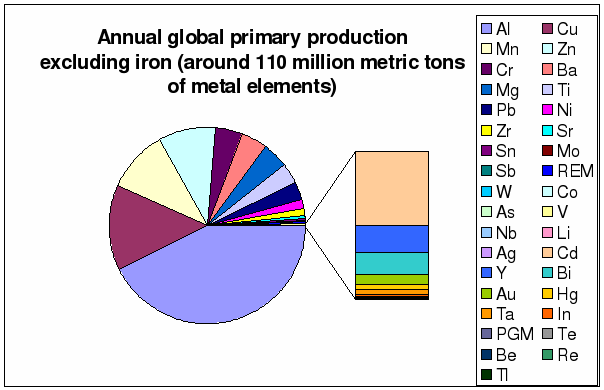
Figure 7: Distribution of annual
global primary production without iron
(based on
table 1)
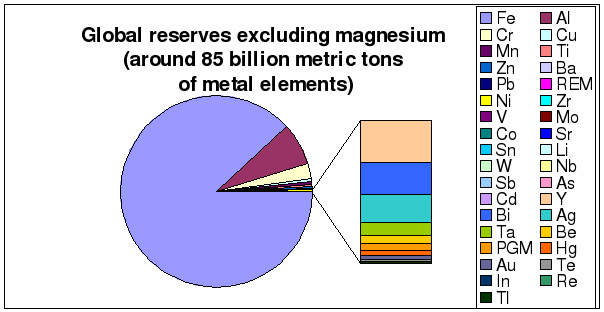
Figure 8: Distribution of global
reserves excluding magnesium (based on table 1)
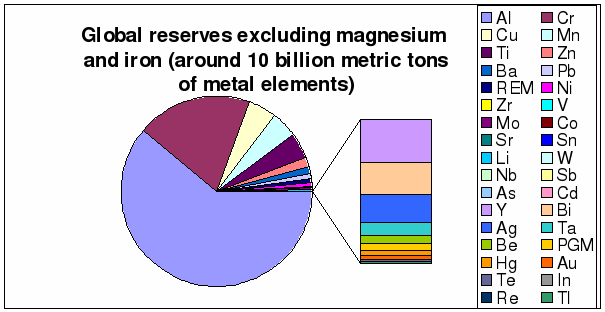
Figure 9: Distribution of global
reserves excluding magnesium and iron
(based on
table 1)
On a trajectory of ‘business as
usual’, we will have much less than 50 years left of cheap and abundant access
to metal minerals. The production rate of metal minerals will start to decline
well in advance of the depletion of reserves as it will take exponentially more
energy input and metal minerals input to grow or even sustain the current
extraction rate of metal minerals. To sustain and increase current production
rates, resources have to be extracted at ever more distant locations (including
deep mining and ocean floor mining) and at ever lower ore grades which require
exponentially more energy to extract. In this sense it could even be stated
that metal minerals scarcity aggravates energy scarcity.
Consequences of unmitigated metal
minerals scarcity
During the next few decades we will
encounter serious problems mining many important metal minerals at the desired
extraction rates. Amongst them are all precious metals (gold, silver and
platinum-group metals), zinc, tin, indium, zirconium, cadmium, tungsten,
copper, manganese, nickel and molybdenum. A number of these metals are already
in short supply (e.g. indium). Metals like gallium, germanium and scandium are
not incorporated in table 1 by lack of data, but these metals suffer from a very
low extraction rate as they are by-products (in very low concentrations) of
other metal minerals; independent production growth is therefore not an option,
thus making an increasing role for these elements impossible.
Besides the
minerals with obvious constraints (low ratio of reserves relative to primary
production), we can distinguish different ‘categories’ of metal minerals in
table 1. First,
several metal minerals which have a high ratio of reserves relative to primary
production suffer from relatively low absolute amounts of reserves and
associated low extraction rates, effectively making them non-viable large-scale
substitutes for other metals which will be in short supply. It is up for debate
for example whether lithium is a viable large-scale substitute for nickel in
accumulators for electric energy as far as land mined lithium is concerned (it
might be extracted from seawater in future [20], albeit at higher cost).
Second, other metal minerals have no acceptable substitutes for their major
applications, which is of special interest for those
metals which will run out relatively fast at the present course, manganese
being an important example. Third, even metals with a high ratio of reserves to
primary annual production combined with large absolute amounts of reserves and
associated extraction rates, can be susceptible to future supply constraints
because they are located in just a few geographic locations. An example is
chromium which is mainly located in
Without timely implementation of
mitigation strategies, the world will soon run out of all kinds of affordable
mass products and services. A few examples are given here. First, a striking
example are cheap mass-produced consumer electronics like mobile phones, flat screen
TVs and personal computers for lack of various scarce metals (amongst others
indium and tantalum). Also, large-scale conversion towards more sustainable
forms of energy production, energy conversion and energy storage would be
slowed down by a lack of sufficient platinum-group metals, rare-earth metals
and scarce metals like gallium. This includes large-scale application of
high-efficiency solar cells and fuel cells and large-scale electrification of
land-based transport. Further, a host of mass-produced products will suffer
from much lower production speeds (or much increased tooling wear) during
manufacturing owing to a lack of the desired metal elements (a.o. tungsten and
molybdenum) for tool steels or ceramics (tungsten carbide). Among the affected
mass-produced machined products are various household appliances and all types
of motorized transport (cars, trains, ships and aero structures). The lack of
various metal elements (a.o. nickel, cobalt, copper) for high-performance
steels and electromagnetic applications will affect all sectors which apply
high-performance rotating equipment. Besides transportation this includes
essential sectors like electric energy generation (coal/oil/gas-based and
nuclear power plants, hydropower, wind power). Also
the vast areas of construction work in general (housing, infrastructure) and
chemical process industries will be affected. The most striking (and perhaps
ironic) consequence of a shortage of metal elements is its disastrous effect on
global mining and primary production of fossil fuels and minerals: these
activities require huge amounts of main and ancillary equipment and consumables
(e.g. barium for barite based drilling mud).
These threats to the global economy
require political, behavioural and governmental activities as well as
technological breakthroughs. Of the breakthroughs, intensified recycling offers
the opportunity to buy us time and innovative substitution may lead to
sustainable options [18,19].
Efficiency: Jevon’s paradox
A potent partial solution for metal
minerals scarcity would be a better extraction
efficiency, if it wasn’t for Jevon’s paradox. Jevon’s paradox is the
proposition that technological progress that increases the efficiency with
which a resource is used, tends to increase (rather than decrease) the rate of
consumption of that resource. So, technological progress on its own (without
‘control’) will only accelerate the depletion of reserves.
Recycling: delaying of effects
Recycling the current and constantly
growing inventory of metal elements in use in various compounds and products is
the obvious choice in order to buy time and avoid or diminish short- to
medium-term supply gaps. Although recycling is nothing new, generally the
intensity could be further enhanced. We should keep in mind though that
recycling has inherent limits, because even 100% recycling (which is virtually
impossible) does not account for annual demand growth. At the present course we
need to continue to expand the amount of metal elements in use in order to satisfy
demand from developing countries like
Substitution: the elements of hope
It is self-evident that - at our
current level of technology - substitution of scarce metals by less scarce
metals for major applications will lead to less effective processes and
products, lower product performance, a loss in product characteristics, or lead
to less environmentally friendly or even toxic compounds. An important and very
challenging task is therefore to realise the desired functionalities of such products
with less scarce elements and to develop processes for production of these
products at an economic scale. The best candidates for this sustainable
substitution are a group of abundantly available elements, that we have
baptised ‘elements of hope’ (see figure 10). These are the most abundant
elements available to mankind and can be extracted from the earth’s crust, from
the oceans and from the atmosphere. They constitute both metal and non-metal
elements. Hydrocarbons for production of materials (including plastics) could
be extracted progressively more from biomass, albeit at a much lower extraction
rate than from concentrated (fossilized) biomass (oil, natural gas and coal).
Not coincidentally, all macronutrients of nature (all flora and fauna including
the human body) are found among the elements of hope: nature either uses these
elements (metabolism, building blocks) or has shown to be tolerant to these
elements (in their abundant natural forms). Substitution based on the elements
of hope therefore is potentially inherently environmentally friendly.
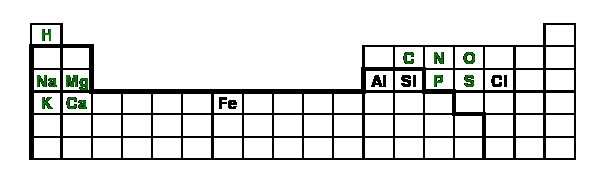
Figure 10: The elements of hope; the
green elements are macronutrients, the elements
within the thickened section are metals
(Si being a metalloid)
Responsible application: frugal and
critical elements
We can look at the remaining global
reserves of metal minerals as a toolbox for future generations (see figure 11).
An important part of the toolbox is reserved for the elements of hope. Another
part of our toolbox is reserved for less abundant but still plentiful building
blocks, the ‘frugal elements’. These elements should only be applied in mass
for applications in which their unique properties are essential. In this way
their remaining reserves will last longer (most notably copper and manganese).
For the sake of completeness, also the non-metals belonging to this category
are included in figure 11. Finally a small corner of the toolbox is reserved
for all other metal elements, the ‘critical elements’, which should be saved
for the most essential and critical applications. Not described in figure 11
but also belonging to the critical elements are other non-metals and the metal
trace elements with high atomic mass (not previously mentioned in this paper by
lack of data from [17]).
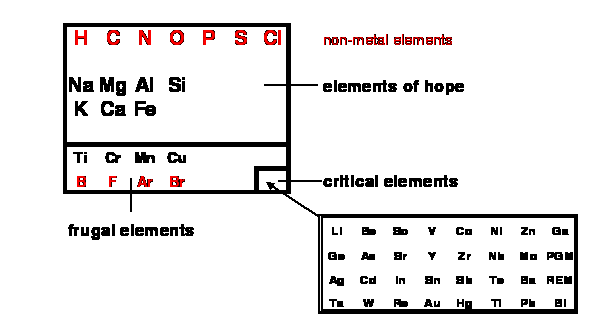
Figure 11: The
toolbox containing the elements of hope, the frugal elements and the critical
elements;
PGM = Platinum-Group Metals;
REM = Rare-Earth Metals;
the red elements are non-metals;
B,Si,Ge,As,Sb,Te are
metalloids
(for a better resolution
version of fig. 11, see this
link )
Conclusion: a call for action,
ingenuity and responsible behaviour
Because of the surging scarcity of
energy, even large-scale substitution and recycling cannot circumvent supply
gaps in metal minerals. This is because production of metals consumes vast
amounts of energy and so do substitution technologies and intensive recycling.
The introduction of managed austerity is required to convince us all to live
using less.
With this paper we call for action. We can increase the lifespan of the
reserves of various materials by making a shift towards large-scale application
of the elements of hope with a sensible use of the frugal and the critical
elements. In order to do this mankind will have to mobilize its collective
creativity and ingenuity. Technology alone is not enough to achieve this goal,
nor can the challenge of metal minerals scarcity be treated as an isolated
problem: it is part of a host of interrelated problems. A solution calls for
nothing less than a globally co-ordinated societal response. The scarcity of
energy, of food and water, of metal minerals and the effects of pollution and
climate change all call for intervention by authorities to facilitate a
transition towards collective responsible behaviour: managed austerity. They
call for a transition from growth in tangible possessions and instant,
short-lived luxuries towards growth in consciousness, meaning and sense of
purpose, connection with nature and reality and good stewardship for the sake
of next generations.
TABLE 1 (Table 1: Primary
production and reserves in metric tons of element content,
based on and derived from [17]])
(for a
higher resolution version, see this
link )
References
[1] Association for the Study of Peak Oil and
gas (ASPO), Newsletter No. 97, compiled by C.J. Campbell, Staball
Hill, Ballydehob, Co. Cork, Ireland, January 2009
[2] Energy Watch Group (EWG), Crude oil -
the supply outlook, EWG-Series No 3/2007,
[3] International Energy Agency, World
Energy Outlook 2008
[4] Koppelaar, R., Meerkerk, B. van, Polder, P.,
Bulk, J. van den, Kamphorst, F., Olieschaarstebeleid (in Dutch),
slotversie, Stichting Peakoil Nederland, October 15, 2008
[5] Simmons, M.R., The energy crisis has
arrived, Energy Conversation Series, United States Department of
Defense, Alexandria, VA, June 20, 2006
[6] The Oil Crunch – Securing the
[7] EWG, Coal: Resources and
Future Production, EWG-Series No 1/2007,
[8] Sousa, L. de, Mearns, E., Olduvai
revisited 2008, posted February 28, 2008 at the website The Oil Drum:
[9] EWG, Uranium Resources and
Nuclear Energy, EWG-Series No 1/2006,
[10] Savinar, M.D., "Are
We 'Running Out'? I Thought There Was 40 Years of the Stuff
Left", http://www.lifeaftertheoilcrash.net, originally published
December 2003,
revised December 2007
[11] Peter, S., Lehmann, H., Renewable
Energy Outlook 2030, Energy Watch Group / Ludwig-Boelkow-Foundation,
November 2008
[12] Wirth, C.J., Peak oil: alternatives,
renewables, and impacts, www.peakoilassociates.com, July 5, 2008.
[13] Bardi, U., Pagani, M., Peak
Minerals, ASPO-Italy and Dipartimento di Chemica
dell’Università di Firenze, posted October 15, 2007 at the website The Oil
Drum:
[14] Meadows, D.,
[15] Skinner, B.J., Exploring
the resource base,
[16] Roper, L.D., Where have all the
metals gone?,
Virginia Polytechnic Institute and
[17] United States Geological Survey
(USGS), Mineral commodity summaries 2008
[18] Bardi, U., The Universal
Mining Machine, posted January 23, 2008 at the website
The Oil Drum
[19] Gordon, R.B., Bertram, M., Graedel, T.E., Metal
Stocks and Sustainability, Proceedings of the National Academy of
Sciences of the U.S., v.103, n.5, January 31, 2006
[20] Bardi, U., Mining the oceans: Can we
extract minerals from seawater?, posted September 22, 2008 at the
website The Oil Drum: